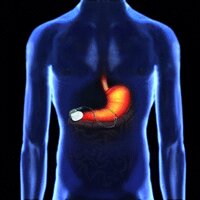
Gastric electrical neurostimulator (GES) - Enterra Therapy Implant
Enterra therapy, like the other treatments is not a cure; it is a method
of managing the condition effectively.
It is a small device that is implanted under the skin, usually in the lower abdominal region. Two insulated wires called leads are implanted in the stomach wall muscle and then connected to the neurostimulator, which then sends mild electrical pulses through the leads to stimulate the smooth muscles of the lower stomach.
First conceived and used in the USA in 1992. Patients there have statistically shown a 50-70% improvement in the frequency of their vomiting episodes.
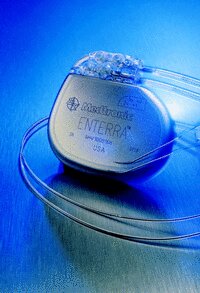
The device is manufactured by Medtronic, who have a UK base - see GP specialists page.
It does not work for all gastroparesis sufferers, and an assessment of an individuals condition would need to be done first before commencing the application process for funding of the surgery.
Some clinical studies have stated that with Enterra therapy:
Findings however are based on limited study groups and other factors such as 'physiological factors' may have affected study outcomes. In conclusion further in depth study is required in order to gain effective results.
See GP Specialists page for further information on clinicians specialising in Enterra Therapy
There are now 150+ patients in the UK being treated using this implant, and 7400+ worldwide (stats July 2012). Mortality rates, with the implant, are low considering the medical complications of those with GP. Deaths are not directly attributable to the implant but to other issues - of the first 10 done, 9 are still alive, and all have required replacement due to battery expiration.
A report based on 188 cases of patients treated with Enterra therapy in the UK, issued by the Scottish Healthboard in March 2012 (see ‘Clinical papers’ page), taking into account statistical information gathered over an extended period, showed that in diabetics there was an 87% reduction in hospital admissions with this therapy, and stated that Enterra therapy to be "statistically significant and clinically important" and has shown across the study group "reductions from baseline in total symptom severity". However proof of cost effectiveness of the treatment remained unproven and a further more detailed, French, 'double blind randomised control study' is to be completed and the results should be published in 2013.
It is a small device that is implanted under the skin, usually in the lower abdominal region. Two insulated wires called leads are implanted in the stomach wall muscle and then connected to the neurostimulator, which then sends mild electrical pulses through the leads to stimulate the smooth muscles of the lower stomach.
First conceived and used in the USA in 1992. Patients there have statistically shown a 50-70% improvement in the frequency of their vomiting episodes.
The device is manufactured by Medtronic, who have a UK base - see GP specialists page.
It does not work for all gastroparesis sufferers, and an assessment of an individuals condition would need to be done first before commencing the application process for funding of the surgery.
Some clinical studies have stated that with Enterra therapy:
- That diabetics are less likely that idiopathic sufferers to achieve relief of bloating symptoms with this treatment.
- That those on opiate type pain relief for their GP symptoms are likely to find Enterra a less effective treatment than the group as a whole.
- Those whose GP cause is due to diabetes or as a post operative complication are more likely to have increased improvement in symptoms than idiopathic sufferers.
- That possibly diabetic sufferers with nausea and vomiting as their main symptoms may be the best candidates for the implant.
Findings however are based on limited study groups and other factors such as 'physiological factors' may have affected study outcomes. In conclusion further in depth study is required in order to gain effective results.
See GP Specialists page for further information on clinicians specialising in Enterra Therapy
There are now 150+ patients in the UK being treated using this implant, and 7400+ worldwide (stats July 2012). Mortality rates, with the implant, are low considering the medical complications of those with GP. Deaths are not directly attributable to the implant but to other issues - of the first 10 done, 9 are still alive, and all have required replacement due to battery expiration.
A report based on 188 cases of patients treated with Enterra therapy in the UK, issued by the Scottish Healthboard in March 2012 (see ‘Clinical papers’ page), taking into account statistical information gathered over an extended period, showed that in diabetics there was an 87% reduction in hospital admissions with this therapy, and stated that Enterra therapy to be "statistically significant and clinically important" and has shown across the study group "reductions from baseline in total symptom severity". However proof of cost effectiveness of the treatment remained unproven and a further more detailed, French, 'double blind randomised control study' is to be completed and the results should be published in 2013.
Temporary GES implants:
Some specialists in the UK do a ‘trial run’ to see whether or not GES will be effective for an individual.
This is done under local anaestetic, and is either by endoscopy or via a small incision into the abdomen and the implant leads are attached to the muscle wall of the stomach, whilst the implant device itself is attached externally.
The patients symptoms are monitored over 3-5 days, either as an in-patient or out patient depending on circumstances.
If the device proves to be effective, following additional stomach emptying tests, then an application for funding for a permanent device will be initiated (see ‘Funding’ below).
Some specialists in the UK do a ‘trial run’ to see whether or not GES will be effective for an individual.
This is done under local anaestetic, and is either by endoscopy or via a small incision into the abdomen and the implant leads are attached to the muscle wall of the stomach, whilst the implant device itself is attached externally.
The patients symptoms are monitored over 3-5 days, either as an in-patient or out patient depending on circumstances.
If the device proves to be effective, following additional stomach emptying tests, then an application for funding for a permanent device will be initiated (see ‘Funding’ below).
The Surgery for a permanent GES implant:
The procedure is relatively quick and straightforward (approximately an hour).
The surgeon makes a small incision, and excavates a ‘pocket’ in the fat layer in which to position the device. The leads are then attached to the implant at one end and the muscles of the stomach wall at the other. A trained technician will then test the device and adjust the settings, prior to the incision being closed.
The incision site is dependent on the surgical team ie. just below breast bone, lower abdomen and is approx 6cm long, dependent on surgical method ie. incision or keyhole.
The procedure is relatively quick and straightforward (approximately an hour).
The surgeon makes a small incision, and excavates a ‘pocket’ in the fat layer in which to position the device. The leads are then attached to the implant at one end and the muscles of the stomach wall at the other. A trained technician will then test the device and adjust the settings, prior to the incision being closed.
The incision site is dependent on the surgical team ie. just below breast bone, lower abdomen and is approx 6cm long, dependent on surgical method ie. incision or keyhole.
Usually as a standard precaution you will spend a short period in ICU
following the surgery before being transferred to a standard ward. And will be
encouraged to ‘get out of bed’ approximately 24 hours post op.
Any post operative pain can be managed by a self administered pain relief intravenous drip, however usually it is more ‘discomfort & soreness’.
Initially only fluids are allowed. It may take some time for you to feel any effects from the implant, but usually it is expected to take affect 48+ hours post op.
Assuming there are no post operative complications, discharge from hospital is usually 5-7 days post op. It is recommend that you have someone with you at this point as your mobility is still limited and, as with all abdominal surgery, you are not supposed to lift weights for a few weeks.
Any post operative pain can be managed by a self administered pain relief intravenous drip, however usually it is more ‘discomfort & soreness’.
Initially only fluids are allowed. It may take some time for you to feel any effects from the implant, but usually it is expected to take affect 48+ hours post op.
Assuming there are no post operative complications, discharge from hospital is usually 5-7 days post op. It is recommend that you have someone with you at this point as your mobility is still limited and, as with all abdominal surgery, you are not supposed to lift weights for a few weeks.

The stitches are usually dissolvable and will not require removal or
intervention.
The implant is not prominent through the skin, and you cannot ‘feel’ it pulsating.
There may be some ongoing pain post op, as the body recovers from surgery and readjusts to eating food. Those that have not eaten solids for prolonged time, often experience this more. But usually the pain eases over a period of a few weeks.
The implant is not prominent through the skin, and you cannot ‘feel’ it pulsating.
There may be some ongoing pain post op, as the body recovers from surgery and readjusts to eating food. Those that have not eaten solids for prolonged time, often experience this more. But usually the pain eases over a period of a few weeks.
Aftercare:
The device can be affected by various 'electrical' equipment, such as electricity pylons, airport x-ray machines, certain x-rays/scans - you may even set off the odd 'anti theft' alarm at your local supermarket!
However you are supplied with an identification card showing your implant serial number and a manual which is easy to follow and explains what you should avoid! When travelling abroad, take the ID card & manual (which is helpfully printed in several languages!) to show at the security gate – you will usually have to undergo a cursory body check as you are unable to go through the scanning machine, however the handheld security wands are fine.
Please note:
Replacement handbooks and ID cards can be issued by the manufacturers Medtronic, upon receipt of a request from your clinician, and must state patient name, address, date of birth, date of implant surgery and facility at which it was done. Requests will not be accepted from individuals or patients.
Special notice should be given to the pages that include advice on 'what to avoid' and precautions to be taken when undergoing other dental, medical or surgical procedures or tests - especially surgical procedures requiring the use of any type of diathermy.
You should have a review appointment with the surgeon approximately 6 weeks post op, to check that the incision is healing properly and that the implant is operating at the correct level.
The device can be affected by various 'electrical' equipment, such as electricity pylons, airport x-ray machines, certain x-rays/scans - you may even set off the odd 'anti theft' alarm at your local supermarket!
However you are supplied with an identification card showing your implant serial number and a manual which is easy to follow and explains what you should avoid! When travelling abroad, take the ID card & manual (which is helpfully printed in several languages!) to show at the security gate – you will usually have to undergo a cursory body check as you are unable to go through the scanning machine, however the handheld security wands are fine.
Please note:
Replacement handbooks and ID cards can be issued by the manufacturers Medtronic, upon receipt of a request from your clinician, and must state patient name, address, date of birth, date of implant surgery and facility at which it was done. Requests will not be accepted from individuals or patients.
Special notice should be given to the pages that include advice on 'what to avoid' and precautions to be taken when undergoing other dental, medical or surgical procedures or tests - especially surgical procedures requiring the use of any type of diathermy.
You should have a review appointment with the surgeon approximately 6 weeks post op, to check that the incision is healing properly and that the implant is operating at the correct level.
Adjustments:
It is common that the device will need to be adjusted/reprogrammed perhaps 2-3 times in the first year and maybe 1-2 each year after. Initial programming of settings, of which they are two: frequency of pulse and length of pulse, may take several adjustments, and therefore some time, to reach the optimum setting for an individual.
This is a straightforward, non invasive procedure, but for legal reasons must be carried out at a registered hospital facility.
Medtronic technicians carry out the adjustments, which in the UK at present (2012) is spread over six centres.
You will know when it requires adjustment as the gastroparesis symptoms return, nausea, vomiting etc - you then contact your surgeon/consultant, who will arrange an appointment for the adjustment clinic. This process can take several weeks as adjustment clinics are usually scheduled at 6-8 week intervals.
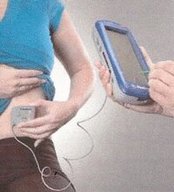
The Medtronic technician holds a small sensor on your abdomen over the implant, and using a remote control, tests the strength of the battery and the frequency, adjusting the strength of pulse and regularity of pulse where necessary. It takes just a few minutes, and the effects can be almost immediate, although for some the symptoms may take a couple of weeks or more to settle. You will be given a printout to keep on your medical records, and the technician keeps a copy.
The device is a sealed unit containing the battery power pack. When the battery 'runs out' the whole device will need replacing ie.another operation. Based on the available USA statistics it is envisaged that the battery has a life expectancy of 8-10 years. But this is dependant on your pacer settings so can vary amongst individuals.
It is common that the device will need to be adjusted/reprogrammed perhaps 2-3 times in the first year and maybe 1-2 each year after. Initial programming of settings, of which they are two: frequency of pulse and length of pulse, may take several adjustments, and therefore some time, to reach the optimum setting for an individual.
This is a straightforward, non invasive procedure, but for legal reasons must be carried out at a registered hospital facility.
Medtronic technicians carry out the adjustments, which in the UK at present (2012) is spread over six centres.
You will know when it requires adjustment as the gastroparesis symptoms return, nausea, vomiting etc - you then contact your surgeon/consultant, who will arrange an appointment for the adjustment clinic. This process can take several weeks as adjustment clinics are usually scheduled at 6-8 week intervals.
The Medtronic technician holds a small sensor on your abdomen over the implant, and using a remote control, tests the strength of the battery and the frequency, adjusting the strength of pulse and regularity of pulse where necessary. It takes just a few minutes, and the effects can be almost immediate, although for some the symptoms may take a couple of weeks or more to settle. You will be given a printout to keep on your medical records, and the technician keeps a copy.
The device is a sealed unit containing the battery power pack. When the battery 'runs out' the whole device will need replacing ie.another operation. Based on the available USA statistics it is envisaged that the battery has a life expectancy of 8-10 years. But this is dependant on your pacer settings so can vary amongst individuals.
Important note regarding adjustments:
For some people it is necessary to adjust the pacer settings several times to achieve the best combination for the individual to reach their optimum benefit from the implant. This takes time as you need several weeks between adjustments to allow the body to adjust.
Many people understandably 'panic' at the first sign of return of symptoms, and book an adjustment clinic appointment immediately and find that their symptoms either settle or that an adjustment is not necessary, thus wasting the appointment for someone in more need. You need to remember that as this device does not cure the condition, and that it is still there in the back ground so to speak, that the GP symptoms may be changing/improving/worsening as a matter of course.
As with anything else many things can affect the digestive system, a bug, unrelated illness, short term medication such as antibiotics, stress, change of enviroment etc - it is important to consider these types of factors, over a period of several weeks, to eliminate them as reasons for symptom return, before booking an adjustment appointment - basically give your body time to adjust to other factors and it may just well settle back to your norm without the need for an adjustment.
It is also important to understand that this device has a limited number of setting combinations and the higher the settings the shorter the battery life - meaning replacement surgery much quicker.
For some people it is necessary to adjust the pacer settings several times to achieve the best combination for the individual to reach their optimum benefit from the implant. This takes time as you need several weeks between adjustments to allow the body to adjust.
Many people understandably 'panic' at the first sign of return of symptoms, and book an adjustment clinic appointment immediately and find that their symptoms either settle or that an adjustment is not necessary, thus wasting the appointment for someone in more need. You need to remember that as this device does not cure the condition, and that it is still there in the back ground so to speak, that the GP symptoms may be changing/improving/worsening as a matter of course.
As with anything else many things can affect the digestive system, a bug, unrelated illness, short term medication such as antibiotics, stress, change of enviroment etc - it is important to consider these types of factors, over a period of several weeks, to eliminate them as reasons for symptom return, before booking an adjustment appointment - basically give your body time to adjust to other factors and it may just well settle back to your norm without the need for an adjustment.
It is also important to understand that this device has a limited number of setting combinations and the higher the settings the shorter the battery life - meaning replacement surgery much quicker.
Statistical Programme:
You may be asked to participate in a post op statistical analysis programme, at regular intervals (3. 6, 12, 24 months and so on).
At present the data collected is kept by the individual specialists and is not collated countrywide. The booklet asks you to note medication, health, nausea, vomiting episodes etc over a two week period.
Funding for the surgery:
In the UK there is no pre approved funding for this procedure, so at present each individual has to submit an application, via their specialist, for special funding to their local ‘Patient care trust’ (PCT) or ‘Health Board’ (HB) and it is up to a panel to decide whether or not to approve each case. Many are refused, and an appeal process can be taken. For further information on funding this surgery please see the Funding for GES page.
In the US legislation has been passed to approve this surgery for the treatment of gastroparesis, and therefore most medical insurance companies and HMO's approve the procedure.
The statistical programme could help to build a UK based picture of the effectiveness of Enterra Therapy - and could ultimately help win the NHS business case to incorporate this device as a standard NHS procedure in the treatment of gastroparesis.
Hopefully in the future a UK wide and in turn Europe wide statistical database will be set up.
Note: (March 2013) - A draft publication by the NHS Commissioning Board 'Clinical Commissioning Policy Statement: Gastric Nerve Stimulation for Gastroparesis' published December 2012 states that GES will not be routinely funded for treatment of diabetic or idiopathic gastroparesis. This document has not yet been formalised as data is still being gathered from specialists in the field. We have been advised that the original NICE guidelines still apply and applications can continue to be made for funding of GES treatment of gastroparesis, pending approval or amendment of this draft proposal.
You may be asked to participate in a post op statistical analysis programme, at regular intervals (3. 6, 12, 24 months and so on).
At present the data collected is kept by the individual specialists and is not collated countrywide. The booklet asks you to note medication, health, nausea, vomiting episodes etc over a two week period.
Funding for the surgery:
In the UK there is no pre approved funding for this procedure, so at present each individual has to submit an application, via their specialist, for special funding to their local ‘Patient care trust’ (PCT) or ‘Health Board’ (HB) and it is up to a panel to decide whether or not to approve each case. Many are refused, and an appeal process can be taken. For further information on funding this surgery please see the Funding for GES page.
In the US legislation has been passed to approve this surgery for the treatment of gastroparesis, and therefore most medical insurance companies and HMO's approve the procedure.
The statistical programme could help to build a UK based picture of the effectiveness of Enterra Therapy - and could ultimately help win the NHS business case to incorporate this device as a standard NHS procedure in the treatment of gastroparesis.
Hopefully in the future a UK wide and in turn Europe wide statistical database will be set up.
Note: (March 2013) - A draft publication by the NHS Commissioning Board 'Clinical Commissioning Policy Statement: Gastric Nerve Stimulation for Gastroparesis' published December 2012 states that GES will not be routinely funded for treatment of diabetic or idiopathic gastroparesis. This document has not yet been formalised as data is still being gathered from specialists in the field. We have been advised that the original NICE guidelines still apply and applications can continue to be made for funding of GES treatment of gastroparesis, pending approval or amendment of this draft proposal.


